Chemistry
Different phases of matter 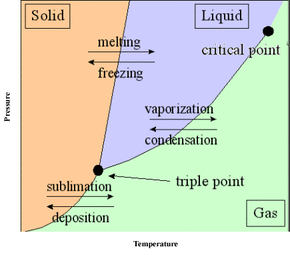 Today, I learned about pressure, temperature, phase changes, and got to use dry ice for the very first time. Pressure and temperature is always used in these phase changes to help the molecules go from one formation to another. Pressure is use to freeze water and gas, and temperature is use to heat solids and water. These are the six different phase changes That I learned:
Today, my teacher give me some dry ice. Dry ice is frozen carbon dioxide, which helps things stay or become frozen. The dry ice was so cold, it kind of burnt my finger. To make dry ice, you must put liquid carbon dioxide inside a tank, then you must lower pressure levels until the liquid forms into a solid.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. Archives
November 2016
Categories |
 RSS Feed
RSS Feed
